The Palatinus Laboratory
The Palatinus Laboratory’s Research: Sudden cardiac death remains the most common cause of natural death in the United States, affecting 300,000-400,000 people per year. A common finding in the hearts of victims of sudden cardiac death is a loss of the normal distribution of ion channels and gap junctions within the heart muscle. A major mechanism behind aberrant localization of these critical ion channels in the diseased heart is a loss of trafficking to their intended location. There are no current therapies that target this protein trafficking defect to treat or prevent arrhythmias or sudden cardiac death. The Palatinus lab is translationally focused on preventing sudden cardiac death by rescuing this protein trafficking defect in the heart. Our lab utilizes a variety of techniques, state-of-the-art high resolution confocal microscopy, molecular biology and live cell imaging as well as transgenic murine models to investigate this trafficking defect. Currently our focus is on targeting this trafficking defect to rescue the cardiomyopathy and sudden death observed in a mouse model of arrhythmogenic cardiomyopathy with the eventual goal to bring these therapeutic strategies to the bedside for preventing sudden cardiac death and treating cardiac arrythmias.
Featured Publications
 | GJA1-20k Rescues Cx43 Localization and Arrhythmias in Arrhythmogenic Cardiomyopathy https://www.ahajournals.org/doi/10.1161/CIRCRESAHA.122.322294 | Connexin43 (Cx43) hemichannels (green) exit the Golgi apparatus and utilize the cytoskeleton (yellow) to traffic to cardiac intercalated discs. In healthy hearts, GJA1-20k (red) organizes the cytoskeleton delivery highway for efficient Cx43 hemichannel trafficking and healthy cardiac rhythm. In mice and humans with Arrhythmogenic Cardiomyopathy, GJA1-20k is diminished, resulting in impaired trafficking highways, limited Cx43 localization to intercalated discs, and ventricular arrhythmia. In a mouse model of Arrhythmogenic Cardiomyopathy, gene therapy with transduction of exogenous GJA1-20k limits cardiac arrhythmia. (Created with Biorender.com) | Joseph A. Palatinus, Steven Valdez, Lindsey Taylor, Claire Whisenant, Craig H. Selzman, Stavros G Drakos, Ravi Ranjan, TingTing Hong, Jeffrey E. Saffitz and Robin M. Shaw22 Feb 2023. Circulation Research. 2023;132:744–746 |
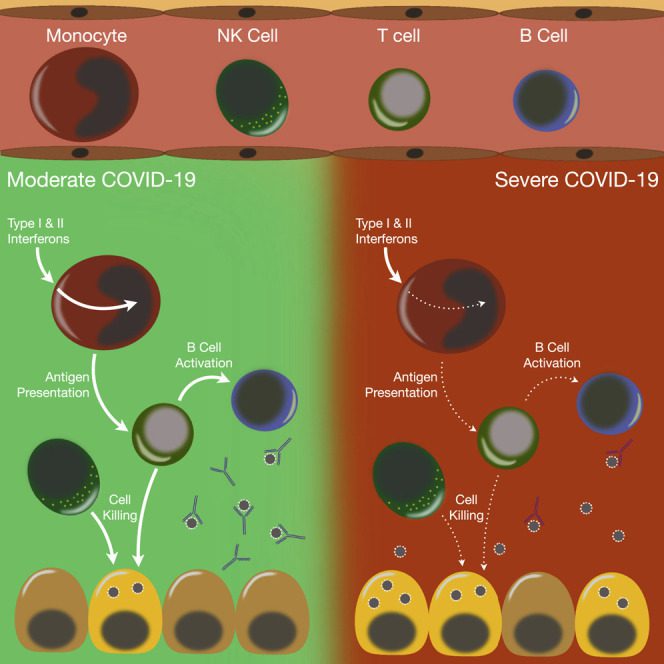 | Cell-Type-Specific Immune Dysregulation in Severely Ill COVID-19 Patients https://pubmed.ncbi.nlm.nih.gov/33357411/ | Our study demonstrates that COVID-19 patients with ARDS have a state of immune imbalance in which dysregulation of both innate and adaptive immune responses may be contributing to a more severe disease course. | Yao, C, Bora, SA, Parimon, T, Zaman, T, Friedman, OA, Palatinus, JA, Surapaneni, NS, Matusov, YP, Cerro Chiang, G, Kassar, AG, Patel, N, Green, CER, Aziz, AW, Suri, H, Suda, J, Lopez, AA, Martins, GA, Stripp, BR, Gharib, SA, Goodridge, HS, Chen, P (2021). Cell-Type-Specific Immune Dysregulation in Severely Ill COVID-19 Patients. Cell Rep, 34, 1:108590. |
 | Interaction of α Carboxyl Terminus 1 Peptide With the Connexin 43 Carboxyl Terminus Preserves Left Ventricular Function After Ischemia-Reperfusion Injury https://pubmed.ncbi.nlm.nih.gov/31422747/ | Conclusions Interaction of αCT1 with the Cx43, but not ZO-1, is correlated with cardioprotection. Pharmacophores targeting Cx43-CT could provide a translational approach to preserving heart function after ischemic injury. | Jiang, J, Hoagland, D, Palatinus, JA, He, H, Iyyathurai, J, Jourdan, LJ, Bultynck, G, Wang, Z, Zhang, Z, Schey, K, Poelzing, S, McGowan, FX, Gourdie, RG (2019). Interaction of α Carboxyl Terminus 1 Peptide With the Connexin 43 Carboxyl Terminus Preserves Left Ventricular Function After Ischemia-Reperfusion Injury. J Am Heart Assoc, 8, 16:e012385. |





