July 2023 Newsletter
Nora Eccles Harrison Cardiovascular Research and Training Institute (CVRTI)
Annual Newsletter | Vol. 2 | July 2023
Welcome to the 2023 Nora Eccles Harrison Cardiovascular Research and Training Institute (CVRTI) Newsletter!
Established in 1967, the CVRTI is a freestanding 40,000 sq. ft. cardiovascular research institute on The University of Utah Health Sciences campus that provides a highly integrated approach to the study of basic and translational cardiac biology. CVRTI personnel include 16 full-time faculty investigators (PhDs, MDs, MD/PhDs, and DVM/PhDs), with over 120 early career investigators (postdocs, graduate students, undergraduates) and dedicated research staff. Our faculty investigators are drawn from three Colleges and six Departments across The University of Utah.
Featured events of this past academic year include continued growth in our extramural funding, a growing list of awards won by our early career investigators and faculty, near completion of the New Research Wing, the addition of Drs. Anthony Donato, Hongchao Guo, and Lisa Lesniewski as new CVRTI Investigators (with recruitment continuing), and Dr. Matt Killeen joins us as Senior Advisor in Translational Research and Entrepreneurship to help us bring our basic discoveries to the bedside as novel therapeutical solutions to cardiovascular disease.
Thank you for taking a moment to review this 2023 progress report.
Robin Shaw, MD, PhD
Director, Nora Eccles Harrison Cardiovascular Research and Training Institute
Nora Eccles Harrison Presidential Endowed Chair
Professor of Medicine, Adjunct Professor of Biomedical Engineering
CVRTI: Basic Advances and Bench to Bedside Solutions
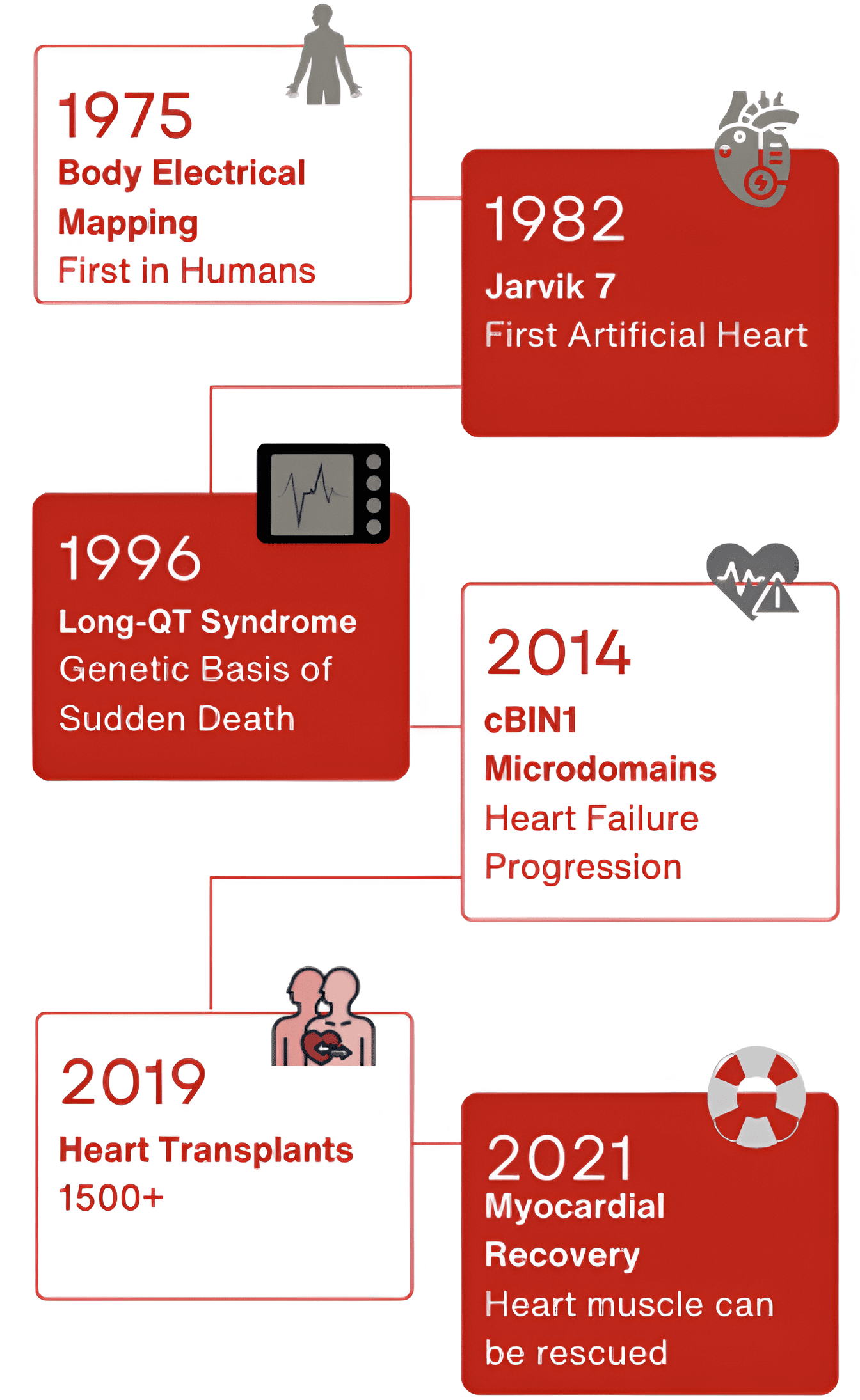
Milestones
Current Activity
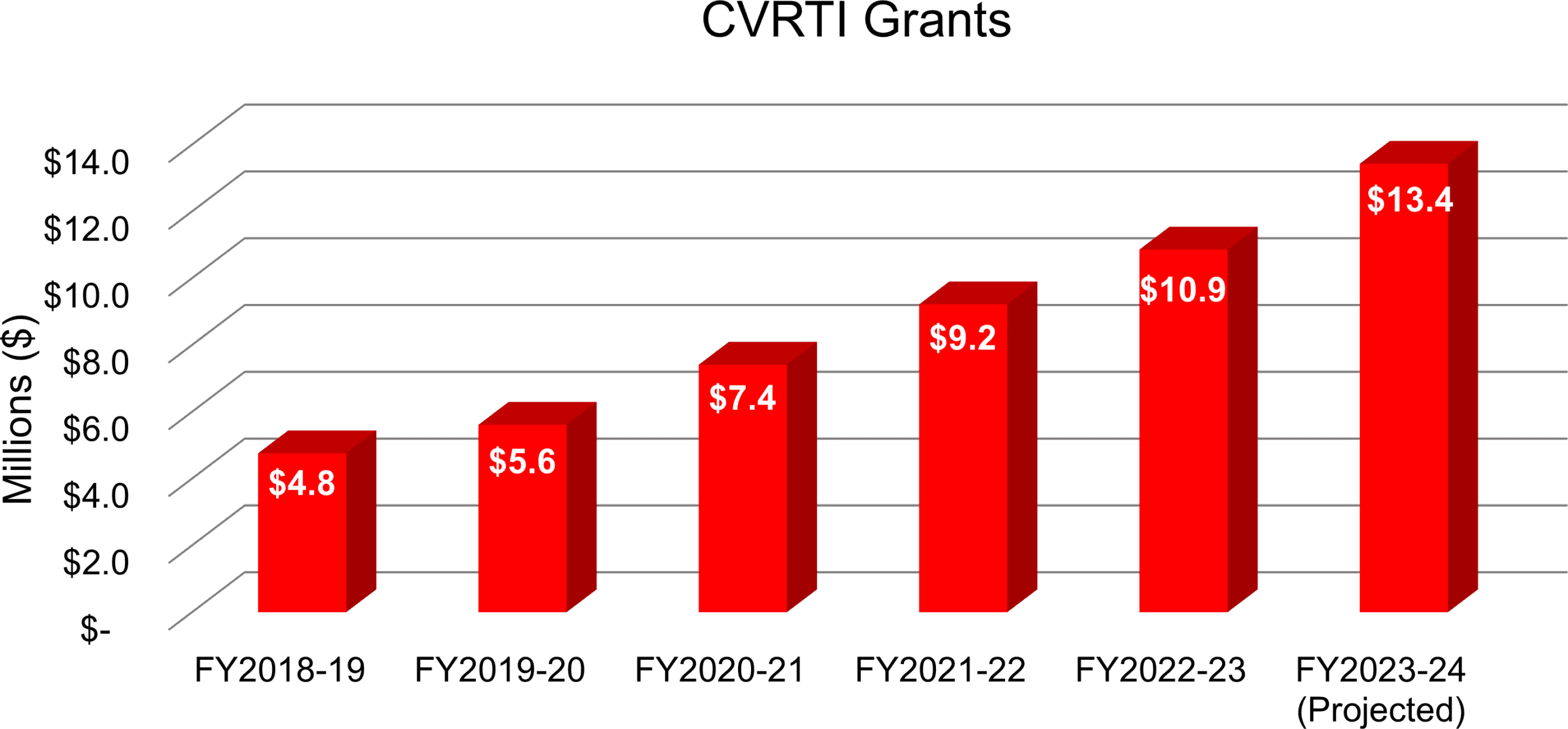
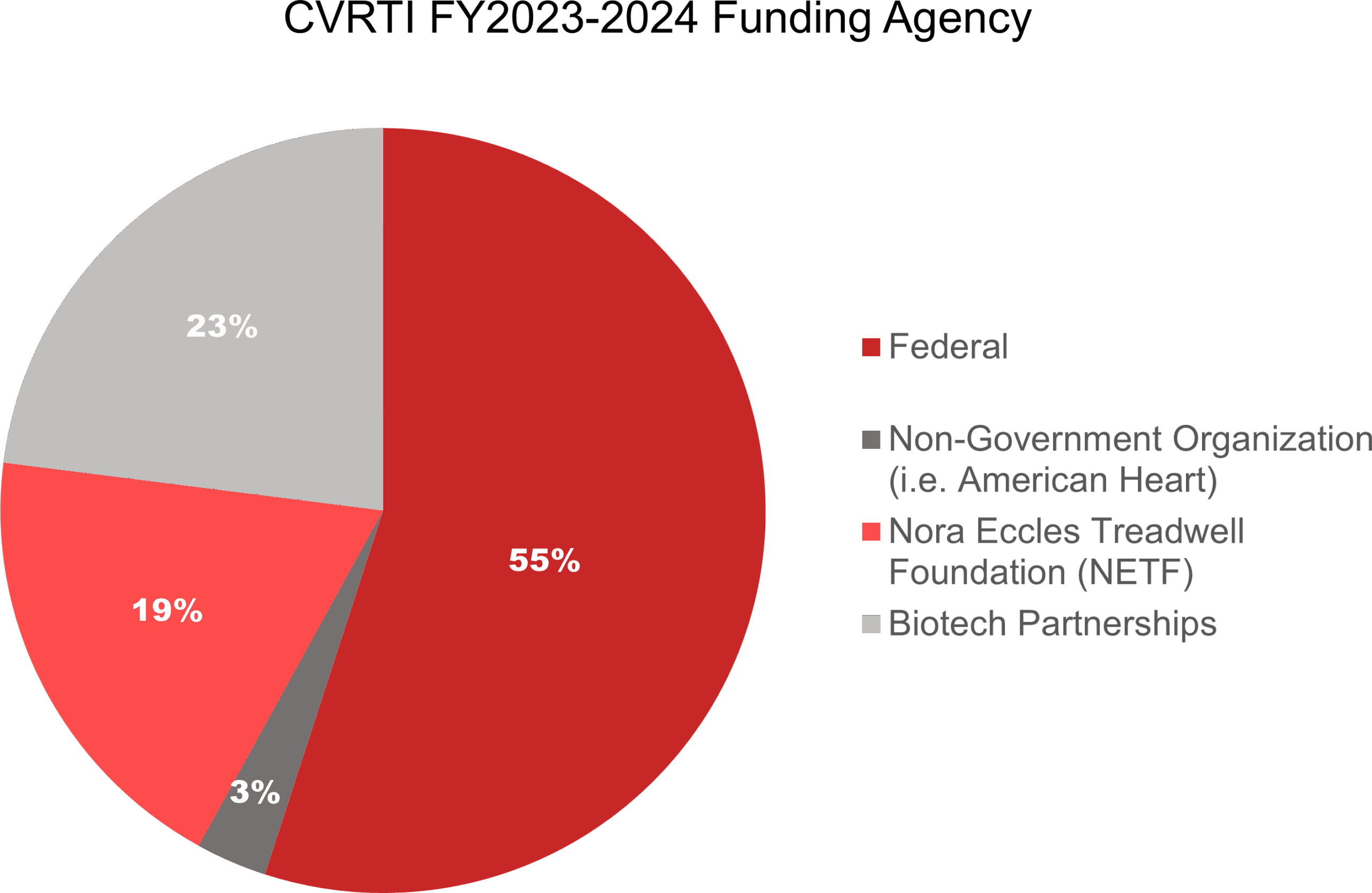
Funding
New Research Wing
The CVRTI footprint includes the addition of a 10,000 sq. ft. New Research Wing generously funded by the Nora Eccles Treadwell Foundation, The University of Utah’s Senior Vice President of Health Sciences, Dr. Michael Good, and The University of Utah’s President Taylor Randall. Construction began in May 2022 with an expected completion date of September 2023 (almost there!). The addition builds upon the strong growth of wet-bench research at the CVRTI and the University as a whole.
The design consists of a new two-story building including wet lab space with additional PI and early career investigator offices, two kitchens, and a gathering space overlooking the majestic Wasatch Mountains. The New Research Wing will also include a mothers’ room on the second floor and a 1,300 sq. ft. freezer farm in the basement.
Time Lapse of Building ConstructionVideo Courtesy of Ken Spitzer, PhD
Follow us on Twitter: @NEH_CVRTI
CVRTI Seminar Series
CVRTI was honored to host extraordinary external speakers for our 2022-2023 Seminar Series. Watch past seminars on our YouTube channel and view the calendar for upcoming speakers on our website!
Our Distinguished External Speakers this past season included:
- Shyam S. Bansal, PhD, The Ohio State University
- Zachary S. Clayton, PhD, University of Colorado Boulder
- Sharlene M. Day, MD, The University of Pennsylvania
- Isabelle Deschenes, PhD, The Ohio State University
- Glenn I Fishman, MD, NYU Grossman School of Medicine
- Shanna Hamilton, PhD, The Ohio State University
- Matthew Killeen, PhD, Renovacor, Inc.
- Javid Moslehi, MD, University of California, San Francisco
- Douglas L. Packer, MD, FAC, HAHA, FHRS, Mayo Clinic, Rochester, Minnesota
- Jeffrey E. Saffitz, MD, PhD, Beth Israel Deaconess Medical Center, Boston, Massachusetts
- Gea-Ny Tseng, PhD, Virginia Commonwealth University
Be sure to join us when the seminar series returns in September, Thursday’s, 12-1 pm MT!
Utah Cardiac Recovery Symposium (U-CARS)
The symposium planning committee was honored to host the 11th Annual Utah Cardiac Recovery Symposium (U-CARS) in March 2023. The U-CARS symposium was hosted in person as well as livestream to participants.
Keynote speaker was:
Christine Seidman, MD
Thomas W. Smith Professor of Medicine and Genetics Director, CV Genetics Center Brigham and Women’s Hospital Harvard Medical School
Featured CVRTI Investigator
Dipayan Chaudhuri, MD, PhD
Dr. Dipayan Chaudhuri earned his medical and graduate degrees at Johns Hopkins University School of Medicine. He then completed a residency in internal medicine at The University of California, San Francisco, and subsequently a clinical fellowship in Cardiovascular Diseases at Massachusetts General Hospital. Concurrently with his clinical fellowship, he completed a postdoctoral research fellowship at Massachusetts General Hospital and Boston Children’s Hospital. In January 2016, Dr. Chaudhuri joined the faculty as an Assistant Professor at The University of Utah, Department of Internal Medicine, Division of Cardiovascular Medicine. He is also an investigator at the Nora Eccles Harrison Cardiovascular Research and Training Institute (CVRTI) on the Health Sciences Campus. His research focuses on changes in metabolism during heart failure. Dr. Chaudhuri is an attending physician on the Cardiovascular Medical Unit and on the Cardiology Consult Service at The University of Utah Hospital, with a clinical specialty in General Cardiology.
Representative Recent Publications
Over the past year, our investigators have published well. Some high impact work in heart failure and metabolism (Drakos and Franklin), vascular biology and senescence (Lesniewski and Donato), and arrhythmia and gene therapy (Palatinus and Shaw) are featured below.
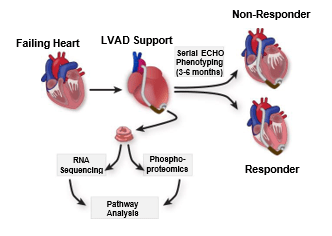
Drakos and Franklin Labs, Circulation, 2023
As a last resort for people with end-stage heart failure, doctors can implant a device called a left ventricular assist device (LVAD), a mechanical pump that takes the load off the heart muscle. While the device is intended to stay in the body permanently, doctors noticed that, in some patients, the assistance provided by the LVAD allowed the patient’s own heart to regain strength and function.
Donato and Lesniewski (TVP Lab), Aging Cell, 2023
Advancing age is a major driver of metabolic dysfunction and diabetes. Elucidating the mechanisms that promote diabetes in older adults is critical to identify novel drugs to treat diabetes. With advancing age, our cells chronically get exposed to damaging agents such as, toxic metabolites, reactive oxygen species, and inflammatory stimuli. These factors promote DNA damage and other dysfunction at the cellular level resulting in a permanent halt of cell division, termed senescence.
Senescent cells promotefurther damage to neighboring healthy cells and thereby amplifying and perpetuate a vicious cycle of local and systemic inflammation. In this study, we found that aging increases senescent cells in metabolic organs such as adipose tissue and the liver. Furthermore, markers of inflammation such as inflammatory cytokines, and abundance of immune cells were elevated in aged adipose tissue and liver compared to young control. In aged mice, we administered a cocktail of senolytic drugs dasatinib and quercetin (D&Q) bimonthly for 3 months. This treatment significantly reduced senescent cells, inflammatory cytokines, and abundance of immune cells in the adipose tissue and liver. This intervention ameliorated glucose and lipid tolerance and suppressed gluconeogenesis, suggesting an improvement in diabetes. D&Q drugs cocktail has been previously approved by Food and Drug Administration to treat chronic kidney disease. Moreover, these drugs have been shown to be effective to treat a host of other age-related pathology. Therefore, we have high hopes that, upon successful clinical trials, D&Q can be used to treat diabetes in older patients.
Palatinus and Shaw Labs, Circulation Research, 2023 (cover)
GJA1-20k rescues Cx43 localization and arrhythmias in arrhythmogenic cardiomyopathy
This report established a new paradigm for arrhythmia treatment: improving Connexin 43 trafficking with gene therapy in arrhythmogenic cardiomyopathy. Here Connexin43 (Cx43) hemichannels (green) exit the Golgi apparatus and utilize the cytoskeleton (yellow} to traffic to cardiac intercalated discs.
In healthy hearts, GJA1-20k (red) organizes the cytoskeleton delivery highway for efficient Cx43 hemichannel trafficking and healthy cardiac rhythm. In mice and humans with Arrhythmogenic Cardiomyopathy, GJA1-20k is diminished, resulting in impaired trafficking highways, limited Cx43 localization to intercalated discs, and ventricular arrhythmia. In a mouse model of Arrhythmogenic Cardiomyopathy, gene therapy with transduction of exogenous GJA1-20k limits cardiac arrhythmia. The results introduce the possibility of treating patients with Arrhythmogenic Cardiomyopathy and other malignant arrhythmia syndromes with a novel and highly efficacious gene therapy.
Click Here to See All CVRTI PublicationsEarly Career Investigator Awards and Honors

Regulation of Purine Metabolism by Protein Methyltransferase Smyd1
Magnus Creed, PhD Candidate
Ruth L. Kirschstein NIH NRSA F31 Predoctoral Fellowship

Targeting EF Hand Domain Containing 1 Protein (EFHD1) in Cardiac Disease
David Eberhardt, PhD
AHA Postdoctoral Fellowship

Exogeneous cBIN1 Gene Therapy Rescues Myocardium in Ischemic Heart Failure
Muhammad S. Khan, PhD
AHA 2023 Career Development Award

Role of Structural Remodeling in Atrial Fibrillation
Eugene Kwan, PhD Candidate
Ruth L. Kirschstein NIH NRSA F31 Predoctoral Fellowship

Personalized Computational Model Based Treatment of Post Ablation Atrial Flutter
Matthias Lange, PhD
AHA Postdoctoral Fellowship

A Novel Mechanism of Mitochondrial Protein Turnover in Complex I Deficient Mitochondrial Cardiomyopathy
Sandra Lee, MD/PhD Fellow
Ruth L. Kirschstein NIH NRSA F30 Predoctoral Fellowship

Adipocyte Enhancer Binding Protein 1 (AEBP1) as a Potential Therapeutic Target to Combat Cardiac Fibrosis
Thirupura S. Shankar, PhD
AHA Predoctoral Fellowship
National Institutes of Health (NIH) Ruth L. Kirschstein Cardiovascular Training Grant (T32) Fellowship

New Insights Into The Pathophysiology Of Dilated Cardiomyopathy In Inherited Phosphoglucomutase I Deficiency
Bijina Balakrishnan, MS, PhD
Evaluating the Effect of Vericiguat on Peripheral Vascular Function, Patient Health Status and Inflammation in Patients with Chronic Heart Failure with Reduced Ejection Fraction
Lina Brinker, MD

cBIN1 Gene Therapy Rescues Ischemic Chronic Heart Failure
Muhammad S. Khan, PhD

Predicting and Understanding Myocardial Recovery in Advanced Heart Failure Patients Undergoing Mechanical Unloading and Circulatory Support
Christos Kyriakopoulos, MD

The Role of Hyaluronan Synthase 2 on Endothelial Glycocalyx with Vascular Aging
Jisok Lim, PhD

The Role of Pyruvate-Lactate Metabolic Axis and Myocardial Salvage Following Ischemic-Reperfusion Injury
Joseph R. Visker, PhD, RCEP

The Role of Hyaluronan Synthase 2 on Endothelial Glycocalyx Targeted DNA-Encoded Libraries for the Discovery of Covalent Sirtuin-6 Activators with Vascular Aging
William K. Weigel, PhD

Regulation of Mitochondrial Health by Mitochondrial Derived Compartments in Cardiomyocytes
Sara Wong, PhD
2022-2023 Dissertation Defenses

Advances in Electrocardiographic Imaging: Technical Improvements and Novel Applications
ake Bergquist, PhD
MacLeod Laboratory, Department of Biomedical Engineering

The Role of DNA Damage, Telomere Dysfunction, and Cellular Senescence in Vascular Aging
Samuel Bloom, PhD
TVP Laboratory, Department of Nutrition and Integrative Physiology

Endothelial Cell ARF6 and mTOR Signaling in Arterial and Metabolic Function
Torikul Islam, PhD
TVP Laboratory, Department of Nutrition and Integrative Physiology

Understanding Mechanisms of Adverse Cardiac Remodeling and Reverse Remodeling in Heart Failure
Thirupura S. Shankar, PhD
Drakos Laboratory, Department of Biomedical Engineering

Utilization of the HIS Bundle for Pacemaker and Implantable Cardioverter Defibrillator Therapy
Annie Hirahara White, PhD
Dosdall Laboratory, Department of Biomedical Engineering
Honors and Recognitions
Vitae 2022, Dipayan Chaudhuri, MD, PHD
Vitae is an event recognizing research excellence across The University of Utah Health Science campus. Dr. Chaudhuri was one of six rising-star faculty who shared stories of their science and how they got to where they are today.
Check out the video below to see Dr. Chaudhuri’s talk “Repairing a Broken Engine: New Approaches to Treat Heart Failure” where he explains how his lab is seeking new therapies to treat mitochondrial dysfunction in heart failure.
Click here to watch videoEmerging Star Award,
Joseph Palatinus, MD, PhD
Dr. Palatinus was recognized for his abstract, GJA1-20k Rescues Connexin 43 Trafficking and Reduces Arrythmias in the Demoglein 2 Mutant Mouse Model of Arrhythmogenic Cardiomyopathy at the International Gap Junction Conference in Spain, July 2022.
Eli S. Gang Most Innovative Abstract Award, Muhammad Khan, PhD
The Heart Rhythm Society awarded Dr. Khan for his abstract, Rescue of Ischemic Dilated Cardiomyopathy in Canines conducted with the Dosdall and Shaw Labs.
Honors and Recognitions
Anthony Donato PhD Professor, Internal Medicine
Dr. Tony Donato received his undergraduate degree from the University of Colorado at Boulder in 1998 and went on to obtain his PhD in exercise physiology from Texas A&M University in 2004. Following his PhD, Dr. Donato completed a postdoctoral fellowship at the University of California, San Diego, and the University of Colorado at Boulder, where he continued his translational focus on vascular physiology and etiology of cardiovascular disease. In 2010, Dr. Donato joined the University of Utah as an Assistant Professor in the Department of Internal Medicine and Division of Geriatrics. As an investigator at the CVRTI and Co-Director of the Translational Vascular Physiology Laboratory at the University of Utah, Dr. Donato’s research focuses on understanding the mechanisms that regulate blood flow and vascular function in health and disease. He has published over 100 peer-reviewed related articles in top-tier scientific journals.
Lisa Lesniewski, PhD
Associate Professor, Internal Medicine
Dr. Lisa Lesniewski received her PhD in kinesiology from Texas A&M University and performed postdoctoral training in metabolism at the University of California, San Diego and vascular aging research at the University of Colorado at Boulder. She is a Professor of Internal Medicine in the Division of Geriatrics at the University of Utah. Dr. Lesniewski co-directs the Translational Vascular Physiology Laboratory and is an investigator at both the CVRTI and the Geriatrics Research Education and Clinical Center at Salt Lake City’s Veterans’ Affairs Medical Center. Her research interest is in elucidating the mechanisms of age-related vascular and metabolic dysfunction and disease. Dr. Lesniewski’s current research seeks to understand the interactions of immune cells and senescence in the metabolic impairments that accompany advancing age, as well as to elucidate the role of the endothelial glycocalyx in age-related vascular dysfunction and disease.
Hongchao Guo, PhD Assistant Professor, Surgery
Dr. Hongchao Guo earned his PhD degree at the University of Nankai (China). He then completed his postdoctoral training at Stanford Cardiovascular Institute mentored by Dr. Joseph Wu. He was promoted to be an instructor at Stanford in 2021. In March 2023, Dr. Guo joined the University of Utah as a tenure-track Assistant Professor at the Department of Surgery. He is also an investigator at the Nora Eccles Harrison Cardiovascular Research and Training Institute (CVRTI) on the Health Sciences Campus. His research focuses on deciphering the genetic mechanisms underlying cardiovascular diseases and cardiac development. His lab employs cutting-edge technologies including CRISPR screening, single-cell sequencing, human 2D and 3D stem cell models, and animal studies to gain broader insights into the genetic understanding of normal and pathological heart function with the ultimate goal of identifying innovative treatments and diagnostics for patients. Dr. Guo has published his research in numerous scientific journals, including Cell, Science Translational Medicine, Development Cell, and Stem Cell Reports. In addition, Dr. Guo has been funded by the National Institute of Health (NIH) K99/R00 career development award. He is also highly supportive on trainees’ career development.
Meet the CVRTI Faculty

Ademuyiwa Aromolaran, PhD

Dipayan Chaudhuri, MD, PhD

Anthony Donato, PhD

Derek Dosdall, PhD

Stavros Drakos, MD, PhD

Sarah Franklin, PhD

Hongchao Guo, PhD

Guillaume Hoareau, DVM, PhD

TingTing Hong, MD, PhD

Lisa Lesniewski, PhD

Rob MacLeod, PhD

Joseph Palatinus, MD, PhD

Ravi Ranjan, MD, PhD

Craig Selzman, MD

Robin Shaw, MD, PhD

Martin Tristani-Firouzi, MD
Meet Dr. Matthew Killeen
Matt Killeen, PhD joined the CVRTI in March 2023 as a Senior Advisor in Translational Research and Entrepreneurship. A key focus of Matt’s role will be partnering with CVRTI faculty to support the translation of groundbreaking scientific discoveries into potentially transformative therapies for cardiovascular diseases, leveraging his extensive experiences in biotechnology R&D, entrepreneurship, and cardiology.
In the biotechnology industry, Matt has played a pivotal role in the discovery and early development of gene and precision therapies for genetic heart diseases, in addition to leading portfolio, R&D, and commercial strategy initiatives for major biotech companies. As Chief Scientific Officer at Renovacor, a publicly traded biotechnology company, Matt led the research organization in the discovery and early development of gene therapies for genetic cardiomyopathies. He developed the R&D strategy, built out the organization, and expanded the pipeline.
He was a key member of the executive team supporting the company’s successful acquisition by Rocket Pharmaceuticals in Dec 2022. Prior to Renovacor, at BioMarin, Matt created the concept for, and founded, the company’s Cardiovascular Therapeutic Area with the mission of leveraging new precision therapy technologies to address the genetic drivers of inherited heart diseases. He built the organization from the ground up, led multiple business development initiatives to in-license new programs, and established a robust precision medicine pipeline, including a gene therapy for MYBPC3 HCM. Previously, Matt led portfolio strategy and strategic planning initiatives at BioMarin and Biogen and was a management consultant for leading biotechnology and pharmaceutical companies.
Matt obtained his PhD in cardiac electrophysiology from the Physiological Laboratory at the University of Cambridge, where his research was focused on arrhythmogenic mechanisms underlying genetic and acquired arrhythmia syndromes. Following this, he was appointed a Research Fellow at Massachusetts General Hospital and Harvard Medical School, before transitioning to the biotechnology industry. Matt has published over 20 scientific articles, authored a textbook on cardiovascular safety during R&D, and has been elected a fellow of the Royal Society of Biology, the American College of Cardiology, and the Heart Rhythm Society.
Meet the Service Core Staff
The CVRTI Core Staff contributes to our productivity and uniquely attractive research environment. It is made up of outstanding individuals, including a facilities manager who supports the research laboratories and coordinates larger projects with The University of Utah’s facilities team; a laboratory specialist, dedicated to the management of CVRTI’s operating room, and an IT specialist. Additionally, we have five individuals responsible for administration including pre-award and post-award grants management, accounting, personnel matters, and coordination of seminar series and events.
Learn More About CVRTI Core StaffCVRTI Events
Staff enjoying the annual summer BBQ, a day on the majestic Utah slopes, and the holiday party at Topgolf
CVRTI is Hiring!
CVRTI is recruiting at all levels, including faculty for our new wing! Click on the Careers button below for specific opportunities and feel free to email CVRTI’s Administrative Manager, Kat Cannon (Kat.Cannon@utah.edu), with any questions.
Careers